
Kelly Grant
Dec. 23, 2020
Health Canada has approved a second COVID-19 vaccine – one developed by Massachusetts-based Moderna, Inc. – making Canada the second country to give the green light to two fully tested vaccines against the pandemic virus.
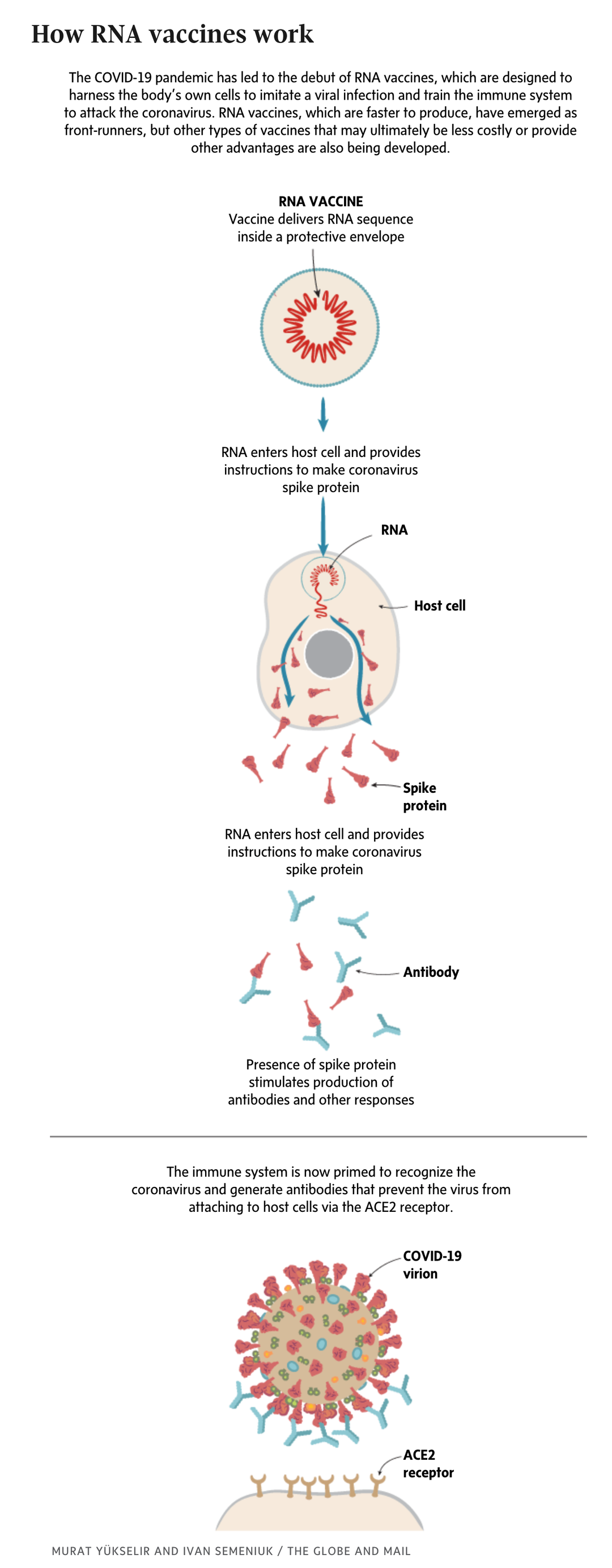
Moderna, a small biotech company, developed its product in co-operation with the U.S. National Institutes of Health. Like the first approved vaccine – from Pfizer, Inc. and BioNTech – Moderna’s shot is an mRNA vaccine. It delivers a snippet of the genetic code of the coronavirus to teach the body what to watch out for, priming the immune system to attack if the real virus comes along. The vaccine does not alter human DNA.
The federal government has secured 40 million doses of Moderna’s vaccine, with an option for another 16 million. The company has promised to deliver 168,000 doses to Canada by the end of December, which should allow the inoculation of more people in long-term care homes and remote Indigenous communities. Moderna’s product is relatively easy to distribute because it can be kept in a regular freezer, unlike Pfizer’s, which must be stored at -70 C.
Here is what we know about the Moderna vaccine.
HOW EFFICACIOUS IS THE VACCINE?
Very. The most recent data from a clinical trial involving about 30,400 people in the United States shows the Moderna vaccine was 94.1-per-cent efficacious at preventing symptomatic COVID-19 two weeks after participants received the second of two doses, which are given 28 days apart.
Researchers tested the vaccine in a randomized, double-blind, placebo-controlled trial, meaning that half the participants got the real vaccine and half were given a placebo. Neither the participants nor the health-care professionals doing the injections knew which was in the syringe.
As trial participants went about their normal lives, 196 of them fell ill with COVID-19, according to the most up-to-date data contained in a briefing document prepared by scientists at the U.S. Food and Drug Administration (FDA). Of those who tested positive, just 11 had received the real vaccine. The rest, 185 people, had been injected with the dummy shot. None of the vaccine recipients suffered a severe case of COVID-19, but 30 of the placebo recipients did, and one of them, a 54-year-old with diabetes, died.
The vaccine was found to be slightly less efficacious – 86 per cent – in people over 65, a group that tends to derive less protection from vaccines because the immune system weakens with age.
Overall, the Moderna efficacy data is “outstanding and exceptionally promising,” said Ilan Schwartz, an assistant professor of infectious diseases at the University of Alberta. That assessment also applies to the Pfizer-BioNTech vaccine, he said.
“People should be getting this vaccine,” Dr. Schwartz said. “Certainly, I’ll be recommending it to all of my family members and I’ll be taking the vaccine without any hesitation.”
IS THE VACCINE SAFE?
Yes, but it’s not without possible side effects. The FDA analysis concluded the vaccine has a “favourable safety profile, with no specific safety concerns” that would prevent the U.S. regulator from granting it what is known as emergency use authorization.
Serious adverse events were rare, but most study participants felt a little sick after being immunized, especially the second time – a sign, experts say, that the vaccine is prodding the immune system into action.
Vaccine recipients reported injection site pain in 91.6 per cent of cases, fatigue in 68.5 per cent, headache in 63 per cent, muscle pain in 59.6 per cent and joint pain in 44.8 per cent.
A larger proportion of participants reported mild and moderate side effects in the Moderna trial than in the Pfizer-BioNTech trial, but Srinivas Murthy, an infectious-diseases doctor at BC Children’s Hospital who specializes in clinical-trial design, cautioned against comparing the two studies. Much depends on how participants are asked about subjective side effects such as muscle pain and headaches, he said. “My muscle pain is not your muscle pain, and how we ask it defines how much is there.”
As in the Pfizer study, a handful of cases of Bell’s palsy, a usually temporary paralysis whose symptoms include the drooping of one side of the face, occurred during the Moderna trial. Three were found in Moderna’s vaccine group and one in the placebo group. Four were found in Pfizer’s vaccine group and none in its placebo group.
Dr. Murthy said the number of Bell’s palsy cases in Moderna’s study was not out of keeping with the number expected in the general population, but regulators should keep a close eye on such cases as the vaccine is rolled out to millions of people.
WHO WAS THE VACCINE TESTED ON?
Moderna’s trial was conducted in the U.S. The company went out of its way to enroll as diverse a population as it could, an effort that delayed the trial and put Moderna behind Pfizer in the race for first approval. Just more than a third of the participants were from racialized communities.
Moderna’s trial also targeted older Americans – a quarter of the participants were over 65 –and people working in essential occupations that put them in the path of the virus. Twenty-five per cent of participants were health care workers.
WERE PREGNANT WOMEN PART OF THE TRIAL?
Not on purpose. Researchers tried to exclude pregnant women, a common and contentious practice in drug development. Female participants were screened for pregnancy and excluded from the study if they tested positive. However, 13 participants wound up pregnant during the trial – six in the vaccine group, seven in the placebo group. They have yet to give birth.
Moderna completed a study involving mating and pregnant rats that found the vaccine “did not have any adverse effects on female reproduction, fetal/embryonal development, or postnatal developmental except for skeletal variations which are common and typically resolve postnatally without intervention,” according to the FDA briefing document.
DOES THE TWO-DOSE VACCINE PROTECT ME AFTER JUST ONE DOSE?
Like the Pfizer-BioNTech vaccine, Moderna’s first shot seems to protect against COVID-19, though not as robustly as two shots given 28 days apart.
In the time between the first and second shot, the vaccine’s overall efficacy was 80.2 per cent. The more time passed, the better. Efficacy was 50 per cent in the 14 days immediately after the first shot and increased to 92.5 per cent in the following two weeks.
The FDA analysis was careful to point out that these partial results do not “provide sufficient information about longer term protection beyond 28 days after a single dose.”
But the first-dose data from Pfizer and Moderna are encouraging enough to have sparked a debate among provinces about the best way to distribute the shots. Some governments are only giving out half of their first allotment and keeping the rest frozen as a way to guarantee that the second doses will be available when they’re needed. Others plan to offer up all of their doses right away and hope the vaccine makers deliver future doses as planned. Proponents of that approach argue that, in the midst of a raging pandemic, it’s best to partially protect as many people as possible as quickly as possible.
DOES THE VACCINE STOP SPREAD OF THE CORONAVIRUS?
Scientists don’t know for certain, but a tantalizing clue in the Moderna trial points to the affirmative.
Neither Moderna nor Pfizer-BioNTech designed their trials to track asymptomatic infections, a task that would have required the frequent swabbing of tens of thousands of study participants. Instead, researchers waited for people to get sick and test positive.
However, Moderna did swab and test everyone for SARS-CoV-2 – the virus that causes COVID-19 – right before injecting them with the second dose of the vaccine or placebo. That effort turned up 52 asymptomatic infections, 38 of them in the placebo group and 14 in the vaccine group. We already know that Moderna’s first dose provides some protection against COVID-19; these findings suggest the vaccine could partly guard against asymptomatic infections, too.
Moderna’s study doesn’t provide a definitive answer to the transmission question, warned Zain Chagla, a McMaster University infectious diseases physician. There were relatively few asymptomatic cases in the study, and participants weren’t tested routinely after their second injection.
This Globe and Mail article was legally licensed by AdvisorStream.
© Copyright 2025 The Globe and Mail Inc. All rights reserved.


